 
There are 2 hydrogen atoms and 1 oxygen atom on the right, so you would write "H=2" and "O=1" under the right side. For the equation H2 + O2 → H2O, there are 2 hydrogen atoms being added to 2 oxygen atoms on the left, so you would write "H=2" and "O=2" under the left side. For example, your equation should look something like "H2 + O2 → H2O." Count the number of atoms in each element on each side of the equation and list them under that side. To balance a chemical equation, first write out your given formula with the reactants on the left of the arrow and the products on the right. If the value you assigned returns coefficients that have a greatest common factor (GCF), simplify the chemical equation by dividing each value by the GCF.If there is only one fraction, multiply all values by that values denominator. If the value you assigned returns fractional values, just multiply all values by the least common multiple (LCM) of the denominators to get rid of the fractions. 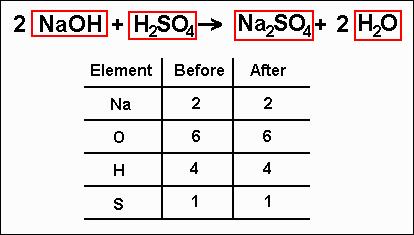
This shows us the values are as follows:.Since H: 2b = 3c + d, we can calculate b like this:.Then start solving the system of equations to get the following values: To quickly do this, take one variable and assign a value to it.You must find the one where every variable is in its smallest, non-fractional form. Since there are more variables than equations, there are multiple solutions. Solve this system of equations to get the numeric value for all the coefficients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
